
Writing Empirical Formulas From Percent Composition - Combustion Analysis Practice Problems - YouTube

The empirical formula of the compound is CHO2 . The molecular weight of the compound is 90. Calculate the molecular formula of the compound.

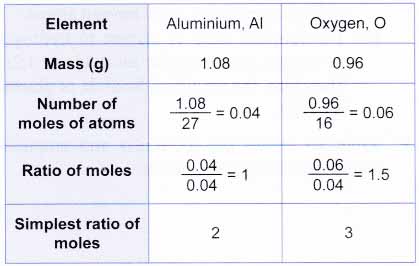
A chemical compound is found to have the following composition : C = 19.5%, Fe = 15.2%, N = 22.8%, K = 42.5% Calculate the empirical formula of the compound. What will



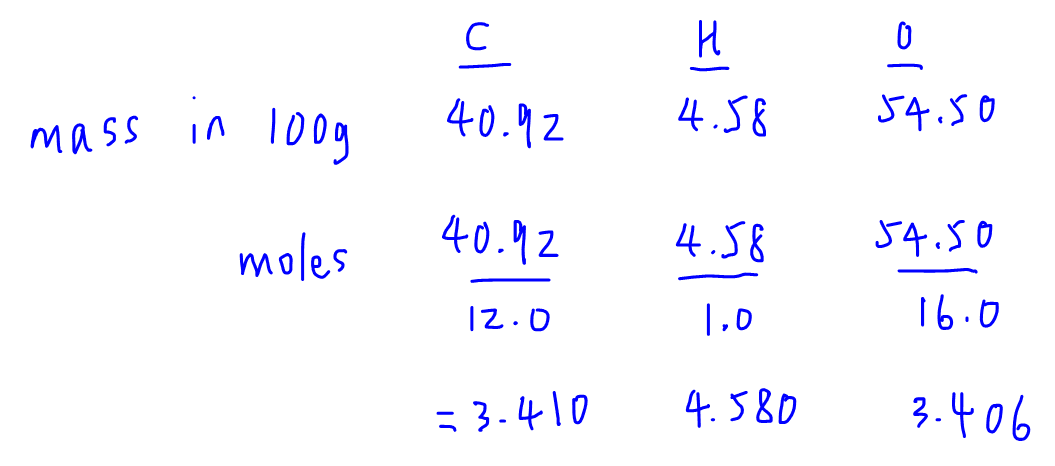

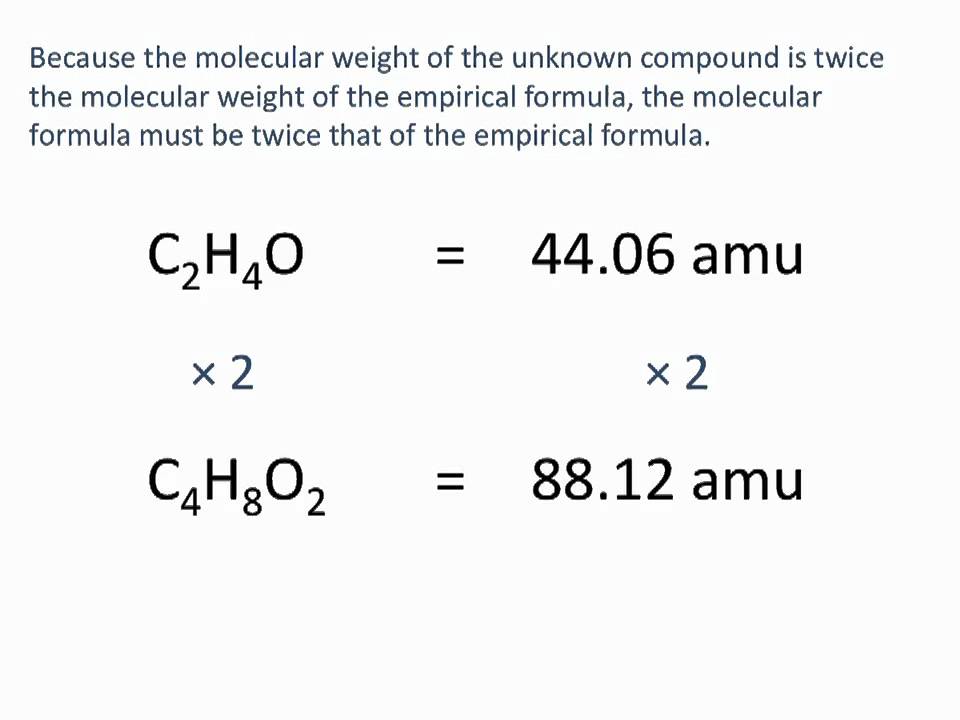






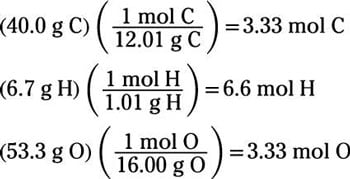

.PNG)





