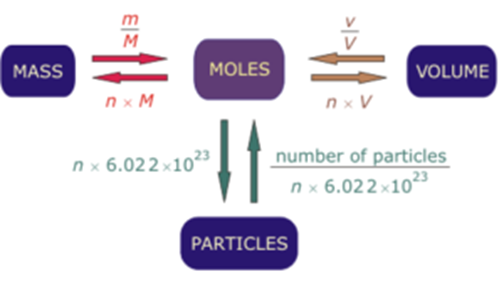
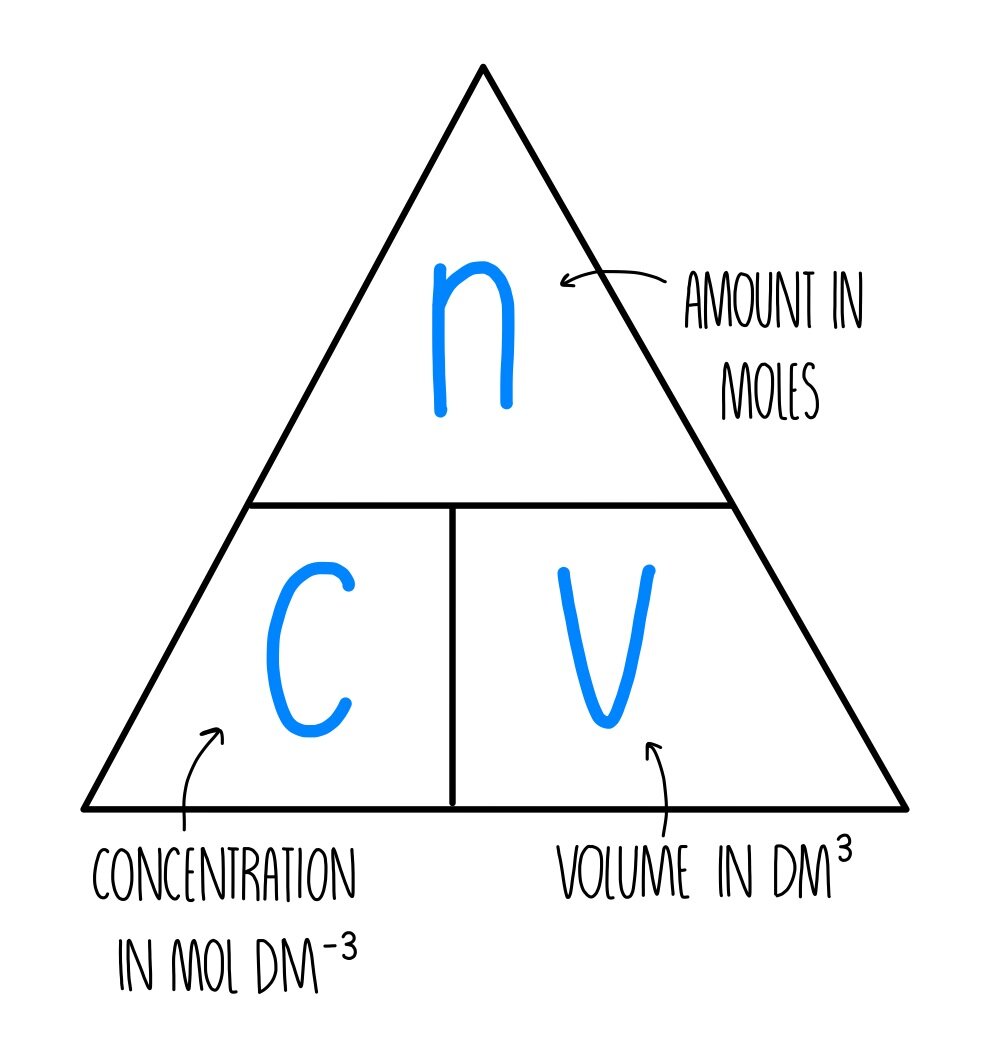
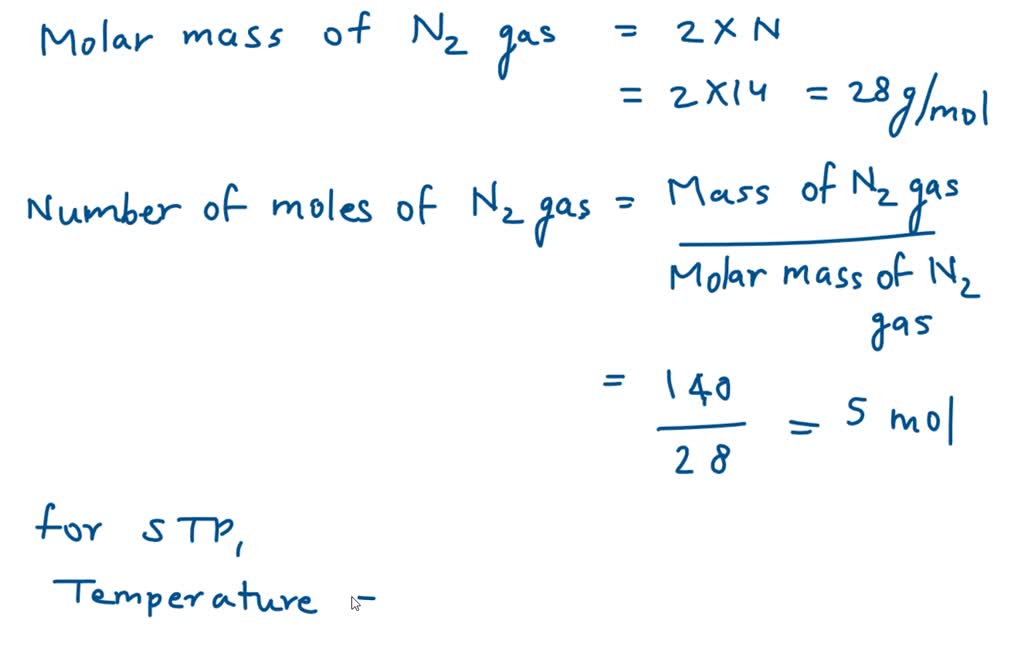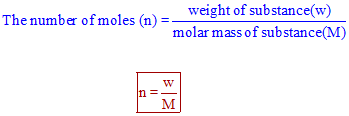Calculate the number of moles for the following: 52 g of He (finding mole from mass) 12.044 × 10^ 23 number of He atoms (finding mole from number of particles)
Calculate the number of moles of atoms present in 56g of nitrogen gas? (If it is written nitrogen gas it should be N2 )
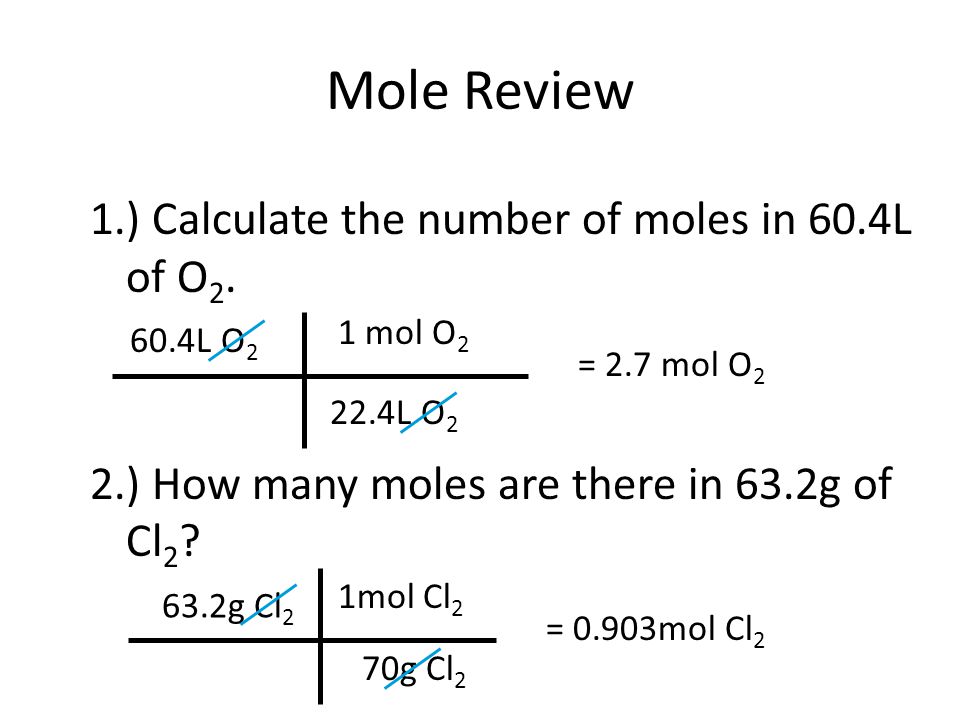
Mole Review 1.) Calculate the number of moles in 60.4L of O2. 2.) How many moles are there in 63.2g of Cl2? 1 mol O2 60.4L O2 = 2.7 mol O2 22.4L
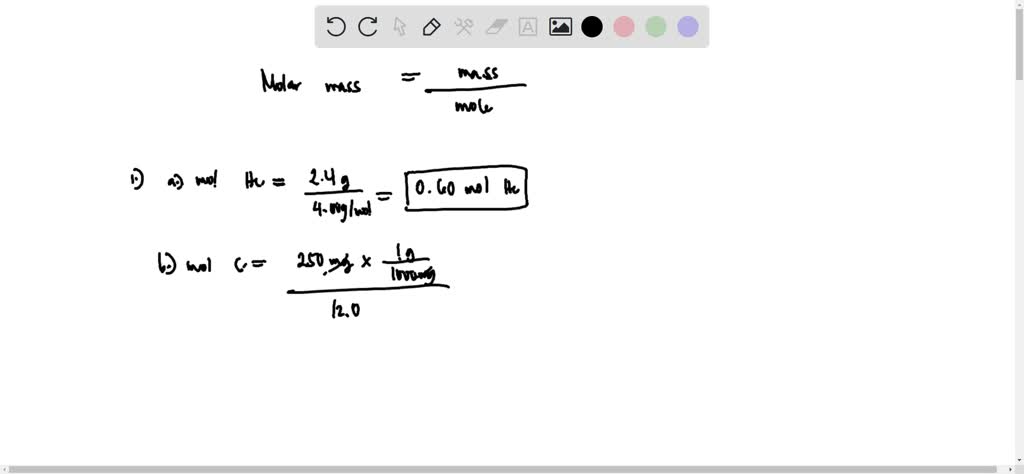
SOLVED: Q1. Calculate the number of moles of each substance in samples with the following masses: a) 2.4 g of He b) 250 mg of Carbon c) 15 g of sodium Chloride

SOLVED: Calculate the number of moles in each sample. a. 72.5 g CCl4 b. 12.4 g C12H22O11 c. 25.2 kg C2H2 d. 12.3 g of dinitrogen monoxide