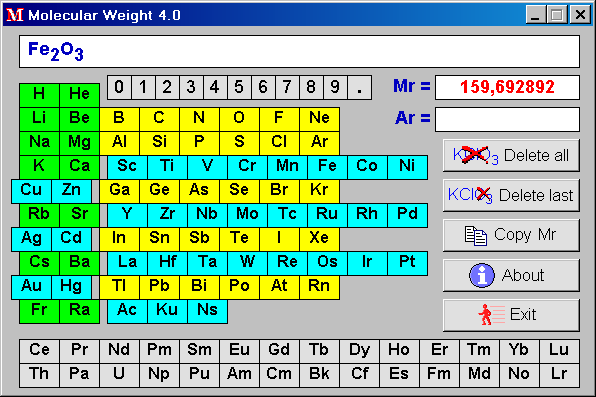
The average molecular mass of a mixture of gas containing nitrogen and carbon dioxide is 36. The mixture contains 280 g of nitrogen. Therefore, the amount of CO2 present in the mixture is :

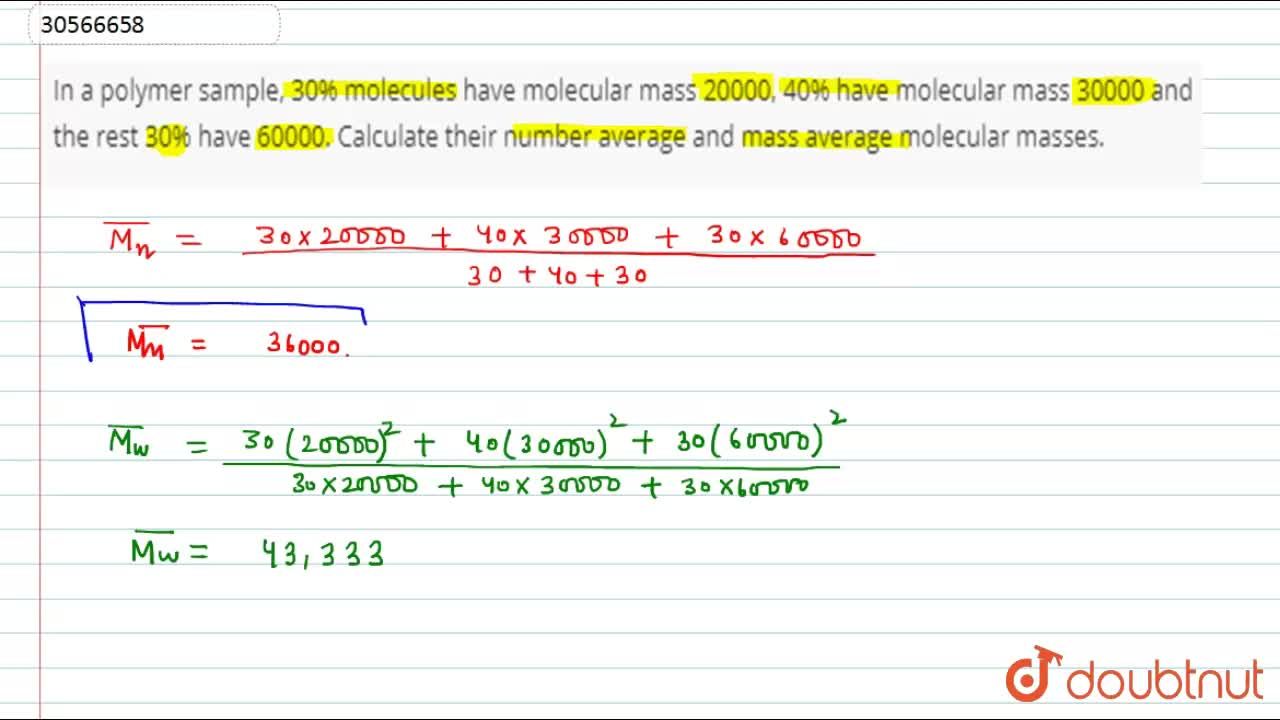
Difference Between Number Average and Weight Average Molecular Weight | Compare the Difference Between Similar Terms









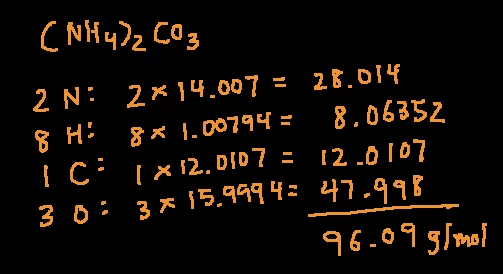


![Potassium Permanganate [KMnO4] Molecular Weight Calculation - Laboratory Notes Potassium Permanganate [KMnO4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/potassium-permanganate-molecular-weight-calculation.jpg)

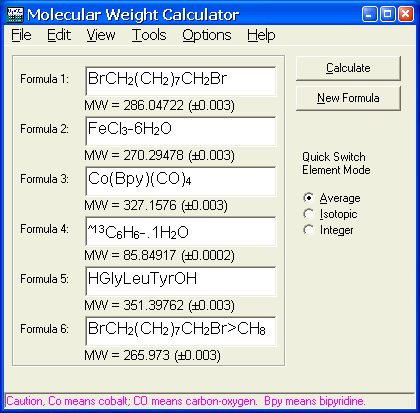
.jpg)